 When I used to teach public relations classes on things like Reputation Management or Crisis Communications, I taught the old PR maxim about “depositing in the bank of goodwill” out there. Simply put, the better you or your organization are at honourable citizenship on a day-to-day basis, the more public goodwill you’ll build up in this account, and the more others will be wiling to trust you.
When I used to teach public relations classes on things like Reputation Management or Crisis Communications, I taught the old PR maxim about “depositing in the bank of goodwill” out there. Simply put, the better you or your organization are at honourable citizenship on a day-to-day basis, the more public goodwill you’ll build up in this account, and the more others will be wiling to trust you.
And vice versa: the more slimy your ongoing behaviour, the less you can realistically expect anybody to trust you. Yes, even when you are telling the truth.
The good news is that, when your balance in the bank of goodwill is healthy, your chances of that trust remaining stable even if you do something bad are improved. So if you should need to make a “withdrawal” one day when a crisis hits, you’ll have the social capital of public trust nicely tucked away in that bank.
It’s also why Phillip Ball – the London-based science journalist, former editor of Nature, and the author of Curiosity: How Science Became Interested in Everything – is taking aim at Big Pharma, and particularly at British drug giant GlaxoSmithKline (GSK). Continue reading

 There is strong evidence that medical researchers’ financial ties to their industry funders may directly influence their published positions in
There is strong evidence that medical researchers’ financial ties to their industry funders may directly influence their published positions in Whenever I feel like I don’t have quite enough aggravation in my life, I used to like checking out
Whenever I feel like I don’t have quite enough aggravation in my life, I used to like checking out  Drug giant AstraZeneca has been working on a back room legal settlement deal with the U.S. government since last fall. And according to a New York Times investigation, the company has earmarked $520 million for the purpose.
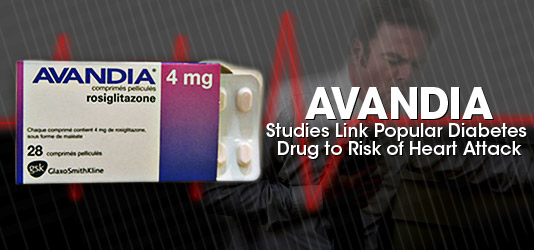
Drug giant AstraZeneca has been working on a back room legal settlement deal with the U.S. government since last fall. And according to a New York Times investigation, the company has earmarked $520 million for the purpose. Poor Toyota. The car maker has been forced to recall more than 8 million vehicles worldwide after news that at least 34 deaths have been linked to Toyota vehicle problems going back as far as 2004. But compare those 34 deaths with the more than 1,000 reports of patient deaths linked with the prescription drug
Poor Toyota. The car maker has been forced to recall more than 8 million vehicles worldwide after news that at least 34 deaths have been linked to Toyota vehicle problems going back as far as 2004. But compare those 34 deaths with the more than 1,000 reports of patient deaths linked with the prescription drug