 A doctor in a white lab coat stands at the pearly gates. The voice of God booms: “And your good deeds?” The man responds: “Well, as a dermatologist, I’ve been warning people that sunlight will kill them and that it’s as deadly as smoking.”
A doctor in a white lab coat stands at the pearly gates. The voice of God booms: “And your good deeds?” The man responds: “Well, as a dermatologist, I’ve been warning people that sunlight will kill them and that it’s as deadly as smoking.”
His smug smile fades as God snaps: “You’re saying that sunlight, which I created to keep you alive, give you vitamin D and make you feel good, is deadly? And the millions of dollars you received from chemical sunscreen companies had nothing to do with your blasphemy?”
A bottle of SPF 1000 sunscreen materializes in the dermatologist’s hand. “You’ll need that where you’re going,” God says. Continue reading →


 Just because a scientific paper sounds authoritative, it doesn’t mean we should always take what’s published in journals as gospel. For example, here’s what scientists might really mean when they pontificate:
Just because a scientific paper sounds authoritative, it doesn’t mean we should always take what’s published in journals as gospel. For example, here’s what scientists might really mean when they pontificate: A doctor in a white lab coat stands at the pearly gates. The voice of God booms: “And your good deeds?” The man responds: “Well, as a dermatologist, I’ve been warning people that sunlight will kill them and that it’s as deadly as smoking.”
A doctor in a white lab coat stands at the pearly gates. The voice of God booms: “And your good deeds?” The man responds: “Well, as a dermatologist, I’ve been warning people that sunlight will kill them and that it’s as deadly as smoking.”
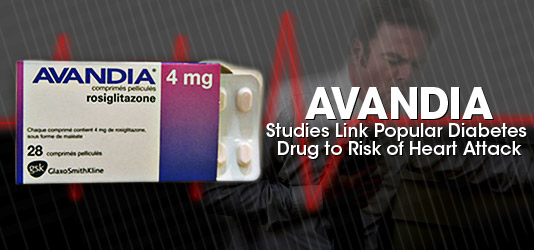
 In case you believe that the medicine you’re taking has been adequately tested on real live patients before being legally approved, you might want to consider research published recently in The New England Journal of Medicine
In case you believe that the medicine you’re taking has been adequately tested on real live patients before being legally approved, you might want to consider research published recently in The New England Journal of Medicine